TSC-ASSOCIATED SEIZURE REDUCTION
EPIDIOLEX®
(cannabidiol)
significantly reduced the frequency of TSC-associated seizures by half
Reduction in monthly frequency of TSC-associated seizures
Results from the 16-week treatment period. Primary endpoint seizures included all countable TSC-associated seizures, including partial-onset† seizures and generalized seizures (tonic-clonic, tonic, clonic, or atonic).
*The median percent reduction from baseline for patients living with TSC was 43% for patients receiving EPIDIOLEX 25 mg/kg/day, compared with 20% for patients receiving placebo (P <0.01).
†Partial-onset seizures (focal) included simple partial seizures (focal motor seizure), complex partial seizures (focal impaired), and secondary generalized tonic-clonic seizures (focal to bilateral tonic-clonic).1
Patients at baseline2:
- Had previously tried a median of 4 prior ASMs
- Currently uncontrolled with a median of 3 current ASMs
89% of patients were taking ≥2 ASMs at baseline and still experiencing 57 TSC-associated seizures per 28 days.2
The most commonly used concomitant ASMs were:
45% valproate|
33% vigabatrin|
29% levetiracetam|
27% clobazam
Reductions in TSC-associated seizure frequency were reported as early as Day 6 in a post hoc analysis of the TSC clinical trial.3
The recommended maintenance daily dosage for EPIDIOLEX is 25 mg/kg/day for patients living with TSC.
Patients with moderate to severe hepatic impairment require a dose adjustment.
TSC-ASSOCIATED PARTIAL-ONSET SEIZURES
EPIDIOLEX®
(cannabidiol)
reduced TSC-associated partial-onset seizure score by 46% in a prespecified exploratory analysis4
Reduction in monthly composite partial-onset seizure score
Results from the 16-week treatment period. Results of the composite seizure score were not controlled for multiplicity; therefore, P-values are not reported.2,4
RESPONDER RATES AND SEIZURE FREEDOM
More patients living with TSC achieved ≥50% and ≥75% reductions in TSC-associated seizures with EPIDIOLEX®
(cannabidiol)
than with placebo
Responder rates (≥50% and ≥75% reductions in TSC-associated seizures from baseline)6
Results from the 16-week treatment period.
More patients achieved freedom from TSC-associated seizures with EPIDIOLEX.
6%
EPIDIOLEX
25 mg/kg/day
0%
PLACEBO
Prespecified exploratory endpoint: Change in mean number of TSC-Associated seizure-free days4*
|
Baseline (per 28 days) |
Maintenance period (per 28 days) |
Percent increase | |
|
EPIDIOLEX 25 mg/kg/day n=71 |
7.4 days | 14.7 days | 97% |
|
PLACEBO n=76 |
7 days | 10.9 days | 55% |
|
EPIDIOLEX 25 mg/kg/day n=71 |
PLACEBO n=76 |
|
|
Baseline (per 28 days) |
7.4 days | 7 days |
|
Maintenance period (per 28 days) |
14.7 days | 10.9 days |
| Percent increase | 97% | 55% |
*Analysis was not controlled for multiplicity and is considered exploratory in nature.4
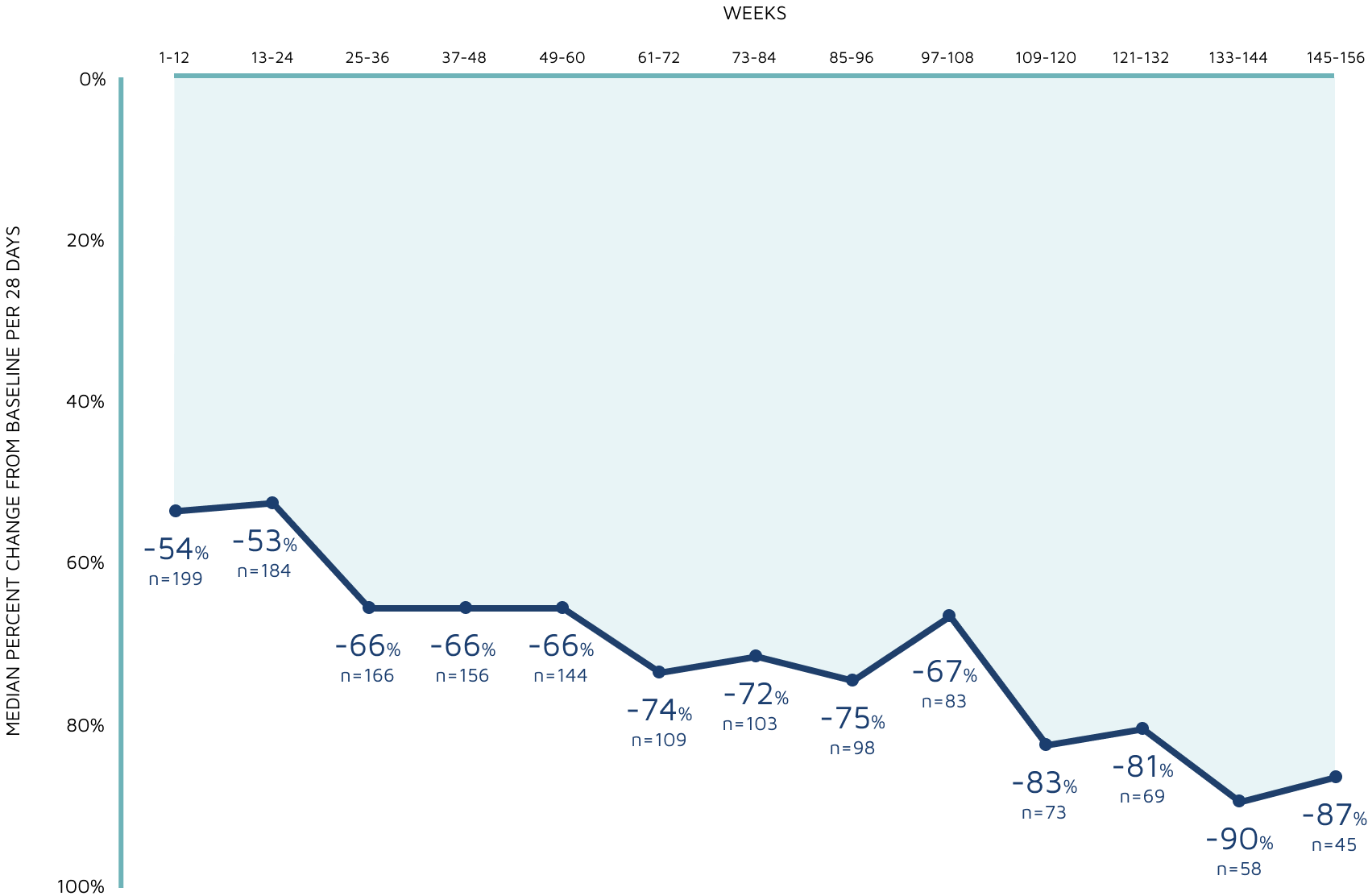
3-YEAR OPEN-LABEL EXTENSION
3-year sustained reduction of TSC-associated seizures7
Open-label extension: Reduction in monthly frequency of TSC-associated seizures7
WEEKS

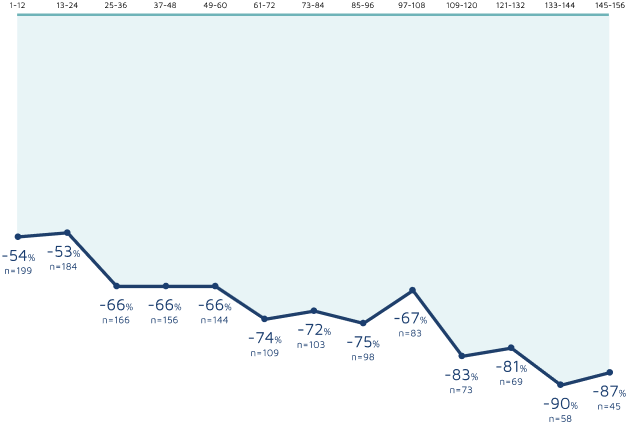
Decreasing n-values reflect a combination of discontinuations and rolling entry into the open-label extension trial.7
- Patients received treatment in the open-label extension for a maximum of 1 year, except in the United States and Poland, where patients could continue beyond 1 year7
- The majority of those eligible transitioned to a commercially available product7
Adverse events:
- The long-term safety profile of EPIDIOLEX®
(cannabidiol)in this open-label extension trial was generally similar to that observed in the EPIDIOLEX clinical development program7
- There was 1 death reported during the study, which was deemed unrelated to treatment by the investigator7
- In the open-label extension trial, titration to doses over 25 mg/kg/day was permitted. At higher doses, an increase in adverse reactions was observed7
Additional resources

Diagnosing an Adult Patient With TSC
A case study of a hypothetical patient with TSC who has been transferred to your care.
Watch now

Patient characteristics
EPIDIOLEX was studied in a wide range of patients living with LGS, Dravet syndrome and TSC, and with concomitant therapies.
See the data

Safety
The safety profile of EPIDIOLEX was evaluated in an expansive clinical trial program.
See the data